Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 , I, I
, I, I , and CsI on C
, and CsI on C fullerene
fullereneKobayashi, Takanori; Yokoyama, Keiichi
Journal of Nuclear Science and Technology, 53(10), p.1489 - 1493, 2016/10
Times Cited Count:6 Percentile:49.29(Nuclear Science & Technology)Theoretical investigation for the adsorption of the cesium atom (Cs), the cesium iodide molecule (CsI), the iodine atom (I), the cesium cation (Cs ), and the iodide anion (I
), and the iodide anion (I ) onto the surface of a single fullerene molecule (C
) onto the surface of a single fullerene molecule (C ) are reported. A hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP) is employed. The adsorption energies, i.e., the opposite of enthalpy change through adsorption, are calculated to be 34, 3, 2, 11, and 12 kcal mol
) are reported. A hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP) is employed. The adsorption energies, i.e., the opposite of enthalpy change through adsorption, are calculated to be 34, 3, 2, 11, and 12 kcal mol for Cs, CsI, I, Cs
for Cs, CsI, I, Cs , and I
, and I , respectively. The equilibrium constant for Cs is calculated to be 7
, respectively. The equilibrium constant for Cs is calculated to be 7 10
10 atm
atm at the temperature of 1000 K and is seven orders of magnitude higher than that for CsI, indicating that the C
at the temperature of 1000 K and is seven orders of magnitude higher than that for CsI, indicating that the C molecule adsorb the Cs atom highly selectively against the CsI molecule.
molecule adsorb the Cs atom highly selectively against the CsI molecule.
 ) by laser ablation in superfluid helium at 1.5K
) by laser ablation in superfluid helium at 1.5KAratono, Yasuyuki; Wada, Akira*; Akiyama, Kazuhiko; Kitazawa, Shinichi; Hojo, Kiichi; Naramoto, Hiroshi*
Chemical Physics Letters, 408(4-6), p.247 - 251, 2005/06
Times Cited Count:1 Percentile:3(Chemistry, Physical)C fulleren was detected in the deposit produced by the laser ablation in superfluid helium at 1.5K. From the mass spectrometry, it was found that the ratio,
fulleren was detected in the deposit produced by the laser ablation in superfluid helium at 1.5K. From the mass spectrometry, it was found that the ratio,  C
C /
/ C
C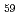
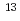 C, differs from statistical one calculated by Poisson distribution for natural graphite. Assuming the recombination of atomic carbons to C
C, differs from statistical one calculated by Poisson distribution for natural graphite. Assuming the recombination of atomic carbons to C molecules as an initial stage for C
molecules as an initial stage for C formation, the different isotopic ratio of C
formation, the different isotopic ratio of C from Poisson statistics was ascribed to the tunneling recombination between
from Poisson statistics was ascribed to the tunneling recombination between  C and
C and 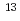 C atoms in the bubble state.
C atoms in the bubble state.
Vacik, J.*; Naramoto, Hiroshi; Kitazawa, Shinichi; Yamamoto, Shunya; Juha, L.*
Journal of Physics and Chemistry of Solids, 66(2-4), p.581 - 584, 2005/02
Times Cited Count:6 Percentile:30.12(Chemistry, Multidisciplinary)no abstracts in English
Sakai, Seiji; Naramoto, Hiroshi; Xu, Y.; Priyanto, T. H.; Lavrentiev, V.; Narumi, Kazumasa
Materials Research Society Symposium Proceedings, Vol.788, p.L11.49.1 - L11.49.6, 2004/00
Mixture films between cobalt and C (CoxC
(CoxC , x = 0.5-700) were prepared by co-evaporation technique under UHV conditions. The composition-dependent features of dilatation and downshift of Raman peak suggest that the mixtures are composed of cobalt particles and a C
, x = 0.5-700) were prepared by co-evaporation technique under UHV conditions. The composition-dependent features of dilatation and downshift of Raman peak suggest that the mixtures are composed of cobalt particles and a C -based phase in which certain number of cobalt atoms are coordinated with C
-based phase in which certain number of cobalt atoms are coordinated with C molecules. It is deduced that the equilibrated number of cobalt atoms in the C
molecules. It is deduced that the equilibrated number of cobalt atoms in the C -based phase is 4 atoms per C
-based phase is 4 atoms per C molecule and in which an electron transfer occurs from a cobalt atom to a C
molecule and in which an electron transfer occurs from a cobalt atom to a C molecule. The evaluation of their temperature dependences reveals that: (1) The mixtures with x
molecule. The evaluation of their temperature dependences reveals that: (1) The mixtures with x 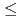 4 are thermally activated-type, and their conductive nature can be attributed to the C
4 are thermally activated-type, and their conductive nature can be attributed to the C -based phase. Further (2) the mixtures between 4
-based phase. Further (2) the mixtures between 4  x
x  60 are also thermally activated, however, the electron hopping process between the isolated cobalt particles is supposed to be operative (variable-range-hopping). And (3) in the mixtures with x
60 are also thermally activated, however, the electron hopping process between the isolated cobalt particles is supposed to be operative (variable-range-hopping). And (3) in the mixtures with x  60 which corresponds to a percolation threshold for cobalt particles, the system changes into metallic conduction.
60 which corresponds to a percolation threshold for cobalt particles, the system changes into metallic conduction.
 molecules in co-deposited film
molecules in co-deposited filmLavrentiev, V.; Abe, Hiroaki*; Yamamoto, Shunya; Naramoto, Hiroshi; Narumi, Kazumasa
Materials Letters, 57(24-25), p.4093 - 4097, 2003/08
Times Cited Count:9 Percentile:38(Materials Science, Multidisciplinary)no abstracts in English
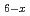 Sr
Sr C
C
Ishii, Kenji; Fujiwara, Akihiko*; Suematsu, Hiroyoshi*; Kubozono, Yoshihiro*
Physical Review B, 65(13), p.134431_1 - 134431_6, 2002/03
Times Cited Count:32 Percentile:78.19(Materials Science, Multidisciplinary)We have studied crystal structure, magnetism and electric transport properties of a europium fulleride Eu C
C and its Sr-substituted compounds, Eu
and its Sr-substituted compounds, Eu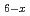 Sr
Sr C
C . They have a
. They have a 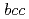 structure, which is an isostructure of other
structure, which is an isostructure of other 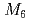 C
C (
(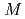 represents an alkali atom or an alkaline earth atom). Magnetic measurements revealed that magnetic moment is ascribed to the divalent europium atom with
represents an alkali atom or an alkaline earth atom). Magnetic measurements revealed that magnetic moment is ascribed to the divalent europium atom with 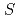 = 7/2 spin, and a ferromagnetic transition was observed at
= 7/2 spin, and a ferromagnetic transition was observed at 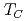 = 10 - 14 K. In Eu
= 10 - 14 K. In Eu C
C , we also confirm the ferromagnetic transition by heat capacity measurement. The striking feature in Eu
, we also confirm the ferromagnetic transition by heat capacity measurement. The striking feature in Eu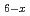 Sr
Sr C
C is very large negative magnetoresistance at low temperature; the resistivity ratio
is very large negative magnetoresistance at low temperature; the resistivity ratio 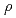 (
(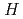 = 9 T)/
= 9 T)/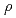 (
(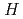 = 0 T) reaches almost 10
= 0 T) reaches almost 10 at 1 K in Eu
at 1 K in Eu C
C . Such large magnetoresistance is the manifestation of a strong
. Such large magnetoresistance is the manifestation of a strong  -
-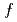 interaction between conduction carriers on C
interaction between conduction carriers on C and 4
and 4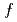 electrons of Eu.
electrons of Eu.
Yoda, Osamu; Miyashita, Atsumi; *; *
JAERI-M 92-173, 27 Pages, 1992/10
no abstracts in English

 irradiation
irradiationKitayama, Takumi*; Morita, Yosuke*; Nakajima, Kaoru*; Suzuki, Motofumi*; Narumi, Kazumasa; Saito, Yuichi; Matsuda, Makoto; Sataka, Masao*; Tsujimoto, Masahiko*; Isoda, Shoji*; et al.
no journal, ,

 from the SiN side, 2
from the SiN side, 2Nakajima, Kaoru*; Marumo, Tomoya*; Nagano, Kengo*; Kimura, Kenji*; Narumi, Kazumasa; Saito, Yuichi
no journal, ,
no abstracts in English
 ions
ionsKitayama, Takumi*; Morita, Yosuke*; Nakajima, Kaoru*; Narumi, Kazumasa; Saito, Yuichi; Matsuda, Makoto; Sataka, Masao*; Kimura, Kenji*
no journal, ,
no abstracts in English
 irradiation
irradiationKitayama, Takumi*; Morita, Yosuke*; Nakajima, Kaoru*; Kimura, Kenji*; Narumi, Kazumasa; Saito, Yuichi; Matsuda, Makoto; Sataka, Masao*
no journal, ,
no abstracts in English
 ion and a single atom heavy ion irradiation
ion and a single atom heavy ion irradiationHirata, Koichi*; Saito, Yuichi; Narumi, Kazumasa; Chiba, Atsuya; Yamada, Keisuke; Matoba, Shiro
no journal, ,
no abstracts in English
 ions
ionsNakajima, Kaoru*; Nagano, Kengo*; Suzuki, Motofumi*; Narumi, Kazumasa; Saito, Yuichi; Hirata, Koichi*; Kimura, Kenji*
no journal, ,
no abstracts in English
Hayashi, Hiroaki*; Kitayama, Takumi*; Morita, Yosuke*; Nakajima, Kaoru*; Narumi, Kazumasa; Saito, Yuichi; Matsuda, Makoto; Sataka, Masao*; Tsujimoto, Masahiko*; Isoda, Shoji*; et al.
no journal, ,
no abstracts in English

 from the backside
from the backsideMarumo, Tomoya*; Nakajima, Kaoru*; Kimura, Kenji*; Narumi, Kazumasa; Saito, Yuichi
no journal, ,
no abstracts in English
 fullerene as the adsorbent of Cs
fullerene as the adsorbent of CsKobayashi, Takanori; Yokoyama, Keiichi
no journal, ,
We have a plan for isotope separation of  Cs using the difference of rotational constants of CsI. In the plan, the adsorbent which can adsorb Cs species-selectively is desirable for the recovery of
Cs using the difference of rotational constants of CsI. In the plan, the adsorbent which can adsorb Cs species-selectively is desirable for the recovery of  Cs after isotope-selective dissociation process of CsI. Paying attention to the difference of the chemical nature of CsI and Cs, fullerenes would be expected to be desirable as the adsorptive material. To confirm the idea, we have calculated the adsorption energy of Cs, CsI etc. to C
Cs after isotope-selective dissociation process of CsI. Paying attention to the difference of the chemical nature of CsI and Cs, fullerenes would be expected to be desirable as the adsorptive material. To confirm the idea, we have calculated the adsorption energy of Cs, CsI etc. to C fullerene using quantum chemistry calculation. We have also calculated the adsorption energy of a cluster model of illite, a kind of minerals which exists in soil and is known as a Cs
fullerene using quantum chemistry calculation. We have also calculated the adsorption energy of a cluster model of illite, a kind of minerals which exists in soil and is known as a Cs adsorbent. The results show that C
adsorbent. The results show that C fullerene would be suitable to adsorb Cs atom in many CsI molecules. In the paper, we will discuss the thermochemical stability of the adducts.
fullerene would be suitable to adsorb Cs atom in many CsI molecules. In the paper, we will discuss the thermochemical stability of the adducts.
Sekiguchi, Tetsuhiro; Yokoyama, Keiichi; Uozumi, Yuki*; Yano, Masahiro; Asaoka, Hidehito; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
For nuclear transmutation of cesium-135 (Cs-135), which is long-lived fission product, we are developing selective absorbent which takes only Cs atom in, but does not CsI. In this study, absorbing property of Cs atom or CsI into fullerene (C ) solid has been investigated using synchrotron-based angle-dependent X-ray photoelectron spectroscopy (ARXPS). It was found that Cs penetrates into C
) solid has been investigated using synchrotron-based angle-dependent X-ray photoelectron spectroscopy (ARXPS). It was found that Cs penetrates into C deep bulk. In contrast, CsI does not diffuse into bulk, although CsI over-layer was formed on the shallow surface. Furthermore, XPS spectra were measured as a function of Ar
deep bulk. In contrast, CsI does not diffuse into bulk, although CsI over-layer was formed on the shallow surface. Furthermore, XPS spectra were measured as a function of Ar -sputtering time in order to know Cs concentration profiles in further deep region. Results showed that Cs penetrates into deep region of several hundreds
-sputtering time in order to know Cs concentration profiles in further deep region. Results showed that Cs penetrates into deep region of several hundreds  .
.